The Law amending and supplementing a number of articles of the Law on Pharmacy has just been passed by the National Assembly with many new points, including regulations on drug trading on e-commerce trading floors...
Delegates press the button to pass the law - Photo: GIA HAN
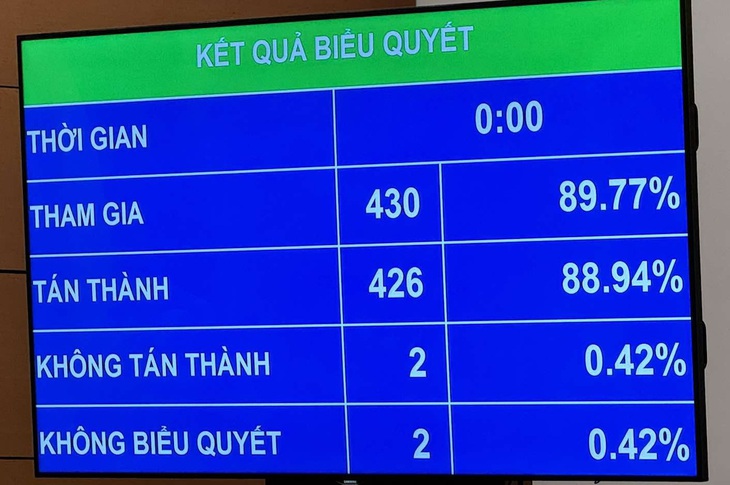
On the afternoon of November 21, with 426/430 delegates participating in the vote, the National Assembly passed the Law amending and supplementing a number of articles of the Law on Pharmacy with 7 important new points. The law will take effect from July 1, 2025, however, some regulations will take effect from January 1, 2025.
Prescription drugs can only be sold online in cases of medical isolation when there is a group A infectious disease
Notably, the new Law on Pharmacy passed by the National Assembly has regulations on pharmaceutical trading via e-commerce.
According to the law, non-prescription drugs can be sold via e-commerce. Prescription drugs can be sold online in cases of medical isolation when there is a group A infectious disease. The law also prohibits online sales of drugs that are subject to special control and drugs on the list of drugs with restricted retail sales.
Established establishments trading drugs and pharmaceutical ingredients via e-commerce must sell on e-commerce trading floors, e-commerce applications, and websites with online ordering functions.
Along with that, the units must comply with the law on advertising and protecting consumer rights; and keep buyers' information confidential.
Units are also required to post certificates of eligibility for pharmaceutical business, pharmaceutical practice certificates, and information on approved drugs.
In addition, drug retail establishments must provide online consultation and instructions on how to use drugs for buyers and deliver drugs to buyers according to detailed instructions from the Minister of Health.
Previously, on behalf of the National Assembly Standing Committee, reporting on the acceptance of explanations, Chairwoman of the Social Committee Nguyen Thuy Anh said that there were opinions proposing specific regulations on the responsibilities of e-commerce trading floors in controlling the quality and origin of drugs sold on the floor.
The Standing Committee of the National Assembly found that the responsibilities of e-commerce trading floors have been stipulated in the law on e-commerce and e-transactions.
In addition, this is only a means to carry out transactions, pharmaceutical business activities will still be carried out by pharmaceutical business establishments.
Therefore, establishments with drugs sold on the floor must be responsible for the quality of the drugs, similar to the traditional form of buying and selling.
The law on e-commerce and e-transactions also stipulates the responsibilities of the parties on the e-commerce trading floor.
The provisions of the draft law are also consistent with the management trend of e-commerce in the pharmaceutical business in a number of countries around the world.
Voting results to pass the law - Photo: GIA HAN
Tightening regulations on drug price management
The recently passed Law on Pharmacy supplements many regulations on drug price management. Accordingly, the Ministry of Health has the right to make recommendations to businesses on drug prices in circulation on the market when they are found to be higher than the highest level of similar types announced, or the selling price in the country of origin.
In addition, the difference between the expected wholesale price of drugs compared to the winning bid price higher than the maximum difference prescribed by the Government is also subject to intervention by the Ministry of Health.
According to the Standing Committee of the National Assembly, the control of non-prescription drug prices is carried out by the method of posting prices for all types of drugs; declaring prices and stabilizing prices for drugs on the List of Essential Drugs.
The announcement of expected wholesale drug prices is a specific measure in drug price management, applied to prescription drugs because this group of drugs accounts for more than 82% of the number of drugs circulating on the market.
These are drugs that account for the majority of use in medical facilities and patients do not buy them for use on their own but must follow the doctor's instructions. This measure helps limit price increases through each level, intermediate level and increase the price when it reaches consumers.

7 NEW POINTS OF THE PHARMACY LAW
The National Assembly Standing Committee said that compared to the current Law, the draft Law has 7 groups of basic new points.
Firstly, the State's policy on pharmaceuticals continues to be perfected, institutionalizing the Party's viewpoint with the goal of developing the Vietnamese pharmaceutical industry into a spearhead industry.
Accordingly, the draft Law adds a number of more breakthrough provisions compared to the 2016 Pharmacy Law to attract investment and further promote research and development of drug and pharmaceutical ingredient production, such as preferential policies on administrative procedures when granting circulation registration certificates and import licenses;
Along with that, the policy applies incentive mechanisms, support from support funds for science and technology activities in research, development, clinical trials, technology transfer, drug and pharmaceutical ingredient production; price maintenance and price reduction policies for some groups of drugs with production technology transfer; digital transformation policies in pharmaceutical activities;
Determine the scale of projects in the pharmaceutical sector that are eligible for incentives, special investment support and assign the Government to specify details to ensure feasibility, bring the State's policies on pharmaceuticals and pharmaceutical industry development into life.
Second, creating a legal corridor for new business forms and methods. That is, the regulation that the establishment of a chain of pharmacies is a separate type of pharmaceutical business establishment, the business conditions, rights and responsibilities of the establishment of a chain of pharmacies, pharmacies in the chain of pharmacies, notably the right to rotate drugs and the right to rotate people responsible for pharmaceutical expertise between pharmacies in the chain of pharmacies;
The Law also regulates the trading of drugs and pharmaceutical ingredients by e-commerce, specifically, supplementing regulations on electronic means, types of drugs and pharmaceutical ingredients allowed to be traded by e-commerce; supplementing the rights and responsibilities of pharmaceutical business establishments by this method.
Third, specify the rights and responsibilities of foreign-invested pharmaceutical businesses in the Law to ensure publicity and transparency in state management. Fourth, expand the rights of manufacturing establishments, export and import establishments, and drug and pharmaceutical ingredient wholesale establishments to sell directly to a number of medical facilities, drug rehabilitation facilities, testing facilities, research and training facilities, and a number of other facilities; allow medical examination and treatment facilities to import drugs to serve the special treatment needs of patients at the medical examination and treatment facility.
Fifth, promote administrative procedure reform in drug and pharmaceutical ingredient circulation registration in the direction of classifying drugs and pharmaceutical ingredients based on different levels of drug properties as well as circulation to adjust records, procedures, time limits for granting, extending, changing, and supplementing drug and pharmaceutical ingredient circulation registration certificates accordingly to increase the ability to access drugs early for people while still ensuring the effectiveness of state management, quality control, safety and effectiveness of drugs; supplement regulations to limit the issuance of duplicate circulation registration numbers.
At the same time, there are specific regulations on dossiers, procedures for registering drugs, drug ingredients, drug testing and drug trading in general to meet the requirements of national defense, security, natural disasters, catastrophes and epidemics.
Sixth, abolish the procedure for confirming drug information content. Strengthen decentralization, delegation of authority, promote the role of the Department of Health in recalling to promptly handle drugs that violate quality in the management area, ensuring safe and effective use of drugs.
Seventh, prescribe price management measures to comply with the Law on Prices and specific measures in drug price management are to announce and re-announce expected wholesale prices applicable to prescription drugs, ensuring that drug wholesale through intermediary levels does not exceed the announced expected wholesale prices.
Mã số thuế/ số CNĐKKD: 0301303865-049
GCNĐĐKKDD số: 5028/ĐKKDD-HCM do SYT TPHCM cấp ngày 16/01/2023
Giấy chứng nhận GPP: 153/GPP cấp ngày 16/01/2023